Periodic Table of Elements - The periodic table is a very useful listing of all 118 elements by symbol, atomic number, and atomic mass and molecular mass. Elements with similar chemical properties are called groups. Visit BYJUS to learn more about it.
Contents
- (a) Chlorine atom (Cl) has atomic number 17. It contains 17 protons and 17 electrons. Chlorine ion (Cl-) is formed when Cl gains one electron. So, Cl-has 18 electrons and 17 protons. Therefore, the electronic configuration of Cl-= 2, 8, 8. (b) Atomic number of Cl-= Number of protons = 17.
- In order to write the Chlorine electron configuration we first need to know the number of electrons for the Cl atom (there are 17 electrons). When we write the configuration we'll put all 17 electrons in orbitals around the nucleus of the Chlorine atom.
- Atomic number and Mass number
- Isotopes
Atom
An atom is the smallest particle of an element which can take part in chemical reaction. Atom consists of three fundamental particles i.e. proton, neutron and electron. Atoms of same elements are similar in properties whereas atoms of different elements are different in properties. Example:- ‘H’ represent the atom of hydrogen.
Proton is positively charged and electron is negatively charged particle. In an atom, number of protons = number of electrons. Hence, the net charge present in an atom is zero i.e. a free atom is chargeless.
Atomic number and Mass number

Atomic number :
- Atomic number is the number of protons present in an atom.
- The modern periodic table is arranged in order of increasing atomic number.
Mass number and Atomic mass :
- Mass number is the sum of the number of protons and the number of neutrons present in an atom. It is a whole number.
Mass no. of an atom = No. of protons + No. of neutrons
- Atomic mass is the average mass of the all of the isotopes of that element. It is a decimal number.
- For example: Hydrogen has three isotopes – 1H1, 1H2 and 1H3 having mass number 1, 2 and 3 respectively. Naturally occurring hydrogen contains about 99.985% of protium, 0.014% of deuterium and 0.001 % of tritium. Therefore the atomic mass of hydrogen is 1.00784 amu.
- The atomic mass of an element element is measured in atomic mass unit (amu, also known as Daltons ‘ D’or unified atomic mass unit ‘u’).
- 1amu = 1.66 x 10-24 grams. 1gm = 6.022 x 1023 amu ( i.e. Avogadro’s number).
Here,
- Atomic number = Number of protons = Number of electrons = 13
- Mass number = No. of protons + No. of neutrons
- No. of neutrons = Mass number – No. of protons = 27-13 = 14.
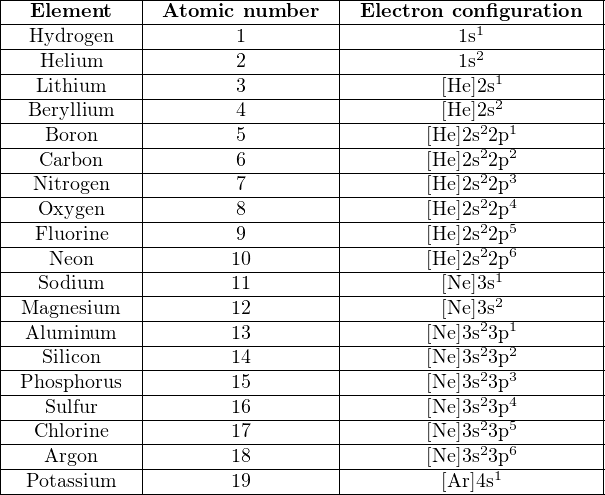
Atomic mass of first 20 elements
| Atomic number | Element | Atomic mass |
| 1 | Hydrogen | 1.008 |
| 2 | Helium | 4.0026 |
| 3 | Lithium | 6.94 |
| 4 | Beryllium | 9.0122 |
| 5 | Boron | 10.81 |
| 6 | Carbon | 12.011 |
| 7 | Nitrogen | 14.007 |
| 8 | Oxygen | 15.999 |
| 9 | Fluorine | 18.998 |
| 10 | Neon | 20.180 |
| 11 | Sodium | 22.990 |
| 12 | Magnesium | 24.305 |
| 13 | Aluminium | 26.982 |
| 14 | Silicon | 28.085 |
| 15 | Phosphorus | 30.974 |
| 16 | Sulfur | 32.06 |
| 17 | Chlorine | 35.45 |
| 18 | Argon | 39.948 |
| 19 | Potassium | 39.098 |
| 20 | Calcium | 40.078 |
Isotopes
Atoms of the same element having same atomic number but different mass number (atomic mass/weight) are called isotopes. For example:
Isotopes of hydrogen :
There are three isotopes of hydrogen:
- Protium or ordinary hydrogen
- Deuterium or heavy hydrogen
- Tritium or radioactive hydrogen.
| Name | Protium | Deuterium | Tritium |
| Symbol | 1H or H | 2H or D | 3H or T |
| No. of protons(P) | 1 | 1 | 1 |
| No. of neutrons(n) | 0 | 1 | 2 |
| No. of electrons(e) | 1 | 1 | 1 |
| Atomic no.(Z) | 1 | 1 | 1 |
| Mass no.(A) | 1 | 2 | 3 |
Naturally occurring hydrogen contains about 99.985% of protium, 0.014% of deuterium and 0.001 % of tritium.
Isotopes have different physical properties since they differ in their mass number.
They have same chemical properties since their electronic configuration is same. However, they differ in the rate of chemical reaction. For example, D2 reacts with Cl2 about 13 times slower than H2 does. The different in rate of reaction due to difference in mass of the atoms of the same element is called isotope effect.
Some other examples of isotopic elements :
| Elements | Isotopes | Most abundant isotope |
| Carbon | 6C12, 6C13, 6C14 | 6C12 |
| Nitrogen | 7N14, 7N15 | 7N14 |
| Oxygen | 8O16, 8O17, 8O18 | 8O16 |
| Sulphur | 16S32, 16S33, 16S34, 16S36 | 16S32 |
| Chlorine | 17Cl35, 17S37 | 17Cl35 |
Isobars
Atoms of different elements having different atomic number but same mass number are called isobars. For example :
18Ar40, 19K40 and 20Ca40
Isotones
Atoms of different elements having different atomic number and mass number but same number of neutrons are called isotones. For example :
6C14, 7N15 and 8O16
Objective questions and their answers
1. Which of the following is known as heavy hydrogen?
a. Protium c. Tritium
b. Deuterium d. Para hydrogen
2. Which of the following is known as radioactive hydrogen?
a. Protium c. Tritium
b. Deuterium d. Para hydrogen
3. Least abundant isotope of hydrogen is:
a. Protium c. Tritium
b. Deuterium d. Heavy hydrogen
4. Diamond and graphite are :
a. Isotopes c. Isotones
b. Isobars d. Allotropes
5. 6C14 and 8O16 are :
a. Isotopes c. Isotones
b. Isobars d. Allotropes
6. 6C14 and 7N14 are :
a. Isotopes c. Isotones
b. Isobars d. Allotropes

7. All particles residing inside the nucleus of an atom are termed as:
a. Protons c. Electrons
b. Neutrons d. Nucleons
8. What makes the atomic mass fractional ?
a.Prerence of isotopes
b. Number of unpaired electrons
c. Spherical shape
d. Quantum number.
Chlorine Atomic Structure
9. Which of the following are not isotopes:
a. 1H1 and 1H3
b. 18K40 and 20Ca40
c. 6C14 and 7N14
d. Both b and c.
10. Charge present in the nucleus of an atom is :
a. Positive c. Chargeless
b. Negative d. Both +Ve and -Ve
11. Molecular weight of heavy water is :
a. 16 c. 20
b. 18 d. 22
Answers :
1. b 2. c 3. c
4. d [Note : different forms of same element having different properties are called allotropes]
5. c 6. b 7. d
8. a 9. d 10. a
11. c Note :Heavy water– Deuterium oxide (D2O) is called heavy water. It’s molecular weight is 20 and boiling paint is 101.50C and melting point is 3.80C.
References
- Sthapit, M.K., Pradhananga, R.R., Foundations of Chemistry, Vol 1 and 2, Fourth edition, Taleju Prakashan, 2005.
| Some tree frogs contain a chlorine compound in their skin that is a very powerful pain killer. This chemical, when used in small doses, has no side effects; in large doses, however, it is fatal. |
Chlorine
| Atomic Number: | 17 | Atomic Radius: | 175 pm (Van der Waals) |
| Atomic Symbol: | Cl | Melting Point: | -101.5 °C |
| Atomic Weight: | 35.45 | Boiling Point: | -34.04 °C |
| Electron Configuration: | [Ne]3s23p5 | Oxidation States: | 7, 6, 5, 4, 3, 2, 1, −1 (a strongly acidic oxide) |
History
From the Greek word chloro, greenish yellow. Discovered in 1774 by Scheele, who thought it contained oxygen. Chlorine was named in 1810 by Davy, who insisted it was an element.
Sources
In nature it is found in the combined state only, chiefly with sodium as common salt (NaCl), carnallite, and sylvite.
Atomic No Of Chlorine Is 17
Properties
It is a member of the halogen (salt-forming) group of elements and is obtained from chlorides by the action of oxidizing agents and more often by electrolysis; it is a greenish-yellow gas, combining directly with nearly all elements. At 10°C one volume of water dissolves 3.10 volumes of chlorine, at 30°C only 1.77 volumes.
Uses
Chlorine is widely used in making many everyday products. It is used for producing safe drinking water the world over. Even the smallest water supplies are now usually chlorinated.
It is also extensively used in the production of paper products, dyestuffs, textiles, petroleum products, medicines, antiseptics, insecticides, food, solvents, paints, plastics, and many other consumer products.
Most of the chlorine produced is used in the manufacture of chlorinated compounds for sanitation, pulp bleaching, disinfectants, and textile processing. Further use is in the manufacture of chlorates, chloroform, carbon tetrachloride, and in the extraction of bromine.
Organic chemistry demands much from chlorine, both as an oxidizing agent and in substitution, since it often brings many desired properties in an organic compound when substituted for hydrogen, as in one form of synthetic rubber.
Chlorine Atomic Symbol
Handling
Chlorine is a respiratory irritant. The gas irritates the mucus membranes and the liquid burns the skin. As little as 3.5 ppm can be detected as an odor, and 1000 ppm is likely to be fatal after a few deep breaths. In fact, chlorine was used as a war gas in 1915.
Atomic No Of Chlorine
Exposure to chlorine should not exceed 0.5 ppm (8-hour time-weighted average - 40 hour week).
